Viral Inactivation Market Influenced by Innovations in Viral Clearance Technologies Enhancing Safety
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Is The Anticipated Rise In Market Value Of The Viral Inactivation Market During The Forecast Period?
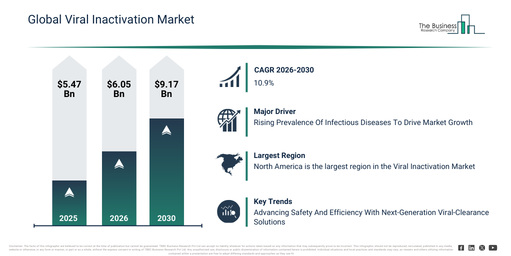
The viral inactivation market size has seen rapid expansion in recent years. It is anticipated to expand from $5.47 billion in 2025 to $6.05 billion in 2026, registering a compound annual growth rate (CAGR) of 10.6%. The historical growth of this market can be attributed to factors like the increasing production of plasma-derived products, the expansion of monoclonal antibody manufacturing, a rising regulatory emphasis on viral safety, the growth of biopharmaceutical manufacturing capacity, and the wider adoption of solvent detergent methods.
The viral inactivation market is projected to experience substantial expansion in the coming years. By 2030, its value is anticipated to reach $9.17 billion, demonstrating a compound annual growth rate (CAGR) of 10.9%. This anticipated growth during the forecast period is primarily driven by factors such as the increasing demand for biologics and biosimilars, growing investments in advanced bioprocessing facilities, an intensified focus on continuous manufacturing, the wider adoption of automated quality control systems, and the expanding pipelines for vaccine production. Key trends emerging over the forecast period encompass the greater uptake of automated viral inactivation systems, an escalating demand for validated virus safety processes, the increased application of continuous flow inactivation methods, the broadening scope of single-use bioprocessing solutions, and a heightened emphasis on process reproducibility and compliance.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=10401&type=smp
What Primary Drivers Are Shaping The Viral Inactivation Market?
The viral inactivation market is projected to expand in the future due to the increasing prevalence of various infectious diseases. These conditions are characterized by their transmission from one person to another, often via contaminated food and beverages, and through pathogens such as viruses, bacteria, fungi, and parasites. Viral inactivation finds extensive application in human disease control initiatives, where physical and chemical disinfectants are utilized to neutralize viruses and prevent the spread of contagious viral infections, thereby fostering the growth of the viral inactivation market. As an illustration, figures from March 2024 released by the Centers for Disease Control and Prevention, a US-based national public health agency, revealed an increase in tuberculosis cases from 8,320 in 2022 to 9,615 in 2023, marking a rise of 1,295 cases. The corresponding rate also rose from 2.5 per 100,000 persons in 2022 to 2.9 in 2023. Thus, the escalating incidence of diverse infectious diseases is a key driver for the growth of the viral inactivation market.
How Are Different Segments Classified In The Viral Inactivation Market Segment Analysis?
The viral inactivation market covered in this report is segmented –
1) By Product: Kits And Reagents, Systems, Services
2) By Method: Solvent Detergent Method, pH Adjustment Method, Pasteurization, Other Methods
3) By Application: Vaccines And Therapeutics, Tissues And Tissue Products, Blood And Blood Products, Other Applications
4) By End User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Blood Banks, Hospital, Academic Research Institutes, Other End Users
Subsegments:
1) By Kits And Reagents: Viral Inactivation Kits, PCR Reagents, Antibodies and Enzymes
2) By Systems: Automated Viral Inactivation Systems, Batch Viral Inactivation Systems, Continuous Flow Systems
3) By Services: Viral Testing Services, Validation Services, Consulting Services
What Trends Are Affecting The Growth Of The Viral Inactivation Market?
Leading enterprises within the viral inactivation market are increasingly focusing on integrating cutting-edge viral-clearance technologies to elevate biologic safety, meet regulatory standards, and boost operational efficiency. These technologies achieve viral inactivation through chemical, physical, or filtration-based approaches, thereby enabling capabilities such as validating log reduction, ensuring regulatory-compliant viral safety, and expediting decision processes in biologics creation. For example, in May 2025, Northway Biotech, a contract development and manufacturing organization (CDMO) based in Lithuania, introduced Full-Service Viral Clearance Studies (VCS). This sophisticated viral-clearance solution is designed to speed up safety validation for protein biologics and gene therapies, incorporating several identical BSL-2 laboratories for efficient high-throughput workflows, BSL-3 testing capabilities, and providing complete regulatory-compliant viral clearance results in less than 10 weeks, which is considerably quicker than typical industry timeframes. This service significantly improves the pace of development, manages costs, and enhances regulatory preparedness for biopharma customers.
Who Are The Companies Driving Activity In The Viral Inactivation Market?
Major companies operating in the viral inactivation market are Merck & Co. Inc., Sartorius AG, Texcell SA, SGS SA, Charles River Laboratories Inc., Clean Cells SAS, Rad Source Technologies, WuXi AppTec, Sigma-Aldrich Co. LLC, Thermo Fisher Scientific Inc., Macopharma, Cerus Corporation, TERUMO BCT INC., Pall Corporation And Eurofins Scientific, Cytiva, Viral Inactivated Plasma Systems SA, Eurofins Scientific SE, Lonza Group AG, Avantor Inc., Bio-Rad Laboratories Inc., F. Hoffmann-La Roche AG
Get The Full Viral Inactivation Market Report:
https://www.thebusinessresearchcompany.com/report/viral-inactivation-global-market-report
Which Region Accounts For The Largest Portion Of The Viral Inactivation Market?
North America was the largest region in the viral inactivation market in 2025. Asia-pacific is expected to be the fastest-growing region in the global viral inactivation market during the forecast period. The regions covered in the viral inactivation market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Viral Inactivation Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/viral-inactivation-global-market-report
Browse Through More Reports Similar to the Global Viral Inactivation Market 2026, By The Business Research Company
Oral Expectorant Market Report 2026
https://www.thebusinessresearchcompany.com/report/oral-expectorant-global-market-report
Antivirals Market Report
https://www.thebusinessresearchcompany.com/report/antivirals-global-market-report
Viral And Non Viral Vector Manufacturing Market Report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model