Clinical Trial Services Market Outlook (2026–2030): Growth Drivers
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Is The Market Size Of The Clinical Trial Services Market Expected To Change From 2026 To 2030?
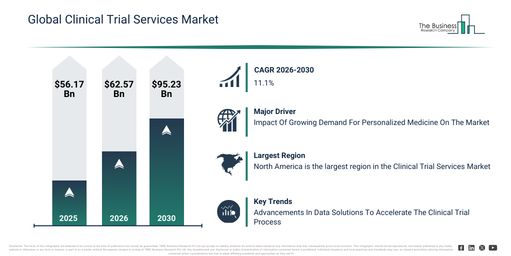
The clinical trial services market has seen considerable expansion in recent times. Its value is projected to increase from $56.17 billion in 2025 to $62.57 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 11.4%. This historical growth is primarily due to higher pharmaceutical R&D expenditure, the broadening of global clinical trial pipelines, an increase in the intricacy of regulatory demands, the rise in multi-therapeutic clinical investigations, and the presence of specialized CRO knowledge.
The clinical trial services market is projected to experience substantial growth in the coming years, with its size anticipated to reach $95.23 billion by 2030, progressing at a compound annual growth rate (CAGR) of 11.1%. This expansion during the forecast period is primarily driven by factors such as the increasing embrace of decentralized and hybrid trial models, a heightened focus on optimizing patient recruitment, the broader integration of real-world evidence, the expanding application of advanced analytics in trial execution, and a growing demand for accelerated trial timelines. Key trends characterizing this period encompass the increased outsourcing of comprehensive clinical trial services, a rising demand for data-centric trial monitoring, the wider adoption of risk-based monitoring frameworks, the expansion of patient-centric trial services, and a strengthened focus on regulatory adherence and compliance assistance.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/report/clinical-trial-services-global-market-report
What Primary Drivers Are Shaping The Clinical Trial Services Market?
The increasing need for personalized medicine is projected to propel the expansion of the clinical trial services market going forward. This medical approach involves customizing treatment and healthcare strategies for individual patients based on their genetic, environmental, and lifestyle factors. Personalized medicine is gaining traction due to advancements in genomic technologies, the rise of targeted therapies, a growing patient desire for tailored treatments, and the integration of AI, biomarkers, and real-world data to improve treatment efficacy and safety. Clinical trial services are crucial for advancing personalized medicine by facilitating the development, testing, and validation of targeted therapies, utilizing genetic data, biomarkers, and customized treatment protocols to ensure more effective and individualized patient outcomes. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based hospital and health care organization, the FDA approved 16 new personalized treatments for rare disease patients in 2023, which was up from six in 2022. Therefore, the escalating demand for personalized medicine is driving the growth of the clinical trial services market.
What Are The Different Segment Types In The Clinical Trial Services Market Segment Breakdown?
The clinical trial services market covered in this report is segmented –
1) By Service Type: Clinical Trial Management (CTM), Planning And Design, Project Management, Monitoring And Data Management, Site Management, Regulatory Services, Regulatory Submission, Consulting, Protocol And Safety Consulting, Other Service Types
2) By Phase: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Area: Oncology, Cardiology, Neurology, Infectious Diseases, Immunology, Respiratory, Dermatology
4) By End-User: Pharmaceutical And Biotechnology Companies, Medical Device Companies, Contract Research Organizations, Academic And Research Institutes
Subsegments:
1) By Clinical Trial Management (CTM): Study Setup And Execution, Recruitment And Patient Enrollment, Trial Monitoring And Reporting, Trial Close-Out And Data Lock
2) By Planning And Design: Protocol Development, Clinical Trial Design, Feasibility Analysis, Site Selection And Initiation
3) By Project Management: Budgeting And Resource Allocation, Risk Management, Timeline Management, Stakeholder Coordination
4) By Monitoring And Data Management: Data Collection And Validation, Statistical Analysis, Quality Control And Assurance, Data Reporting And Interpretation
5) By Site Management: Site Initiation And Training, Site Monitoring And Audits, Site Close-Out And Documentation
6) By Regulatory Services: Regulatory Compliance And Consultation, Regulatory Strategy Development, Preparation For Audits And Inspections
7) By Regulatory Submission: Submission Of Investigational New Drug (IND) Applications, New Drug Application (NDA) Submission, Submission Of Clinical Study Reports (CSRs)
8) By Consulting: Strategy And Planning Consulting, Operational And Performance Consulting, Data Analytics And Interpretation Consulting
9) By Protocol And Safety Consulting: Clinical Trial Protocol Consulting, Safety Management And Risk Assessment, Safety Reporting And Adverse Event Management
10) By Other Service Types: Medical Writing, Biostatistics And Data Analysis Services, Patient Recruitment Services, Post-Approval Studies
What Trends Are Affecting The Growth Of The Clinical Trial Services Market?
Major companies operating in the clinical trial services market are increasingly focusing on developing sophisticated data solutions to expedite the clinical trial process. Data solutions refer to various technologies and tools that effectively manage, analyze, and optimize data to significantly improve efficiency, accuracy, and informed decision-making in critical processes like clinical trials. As an illustration, in July 2024, Laboratory Corporation, a US-based life sciences company, launched Labcorp Global Trial Connect, a comprehensive set of central laboratory solutions aimed at streamlining and enhancing the entire clinical trial process. This integrated platform facilitates seamless communication and real-time data sharing by connecting central laboratories with clinical trial sites and study sponsors. This approach helps to optimize the clinical trial process, reducing potential delays and improving overall trial timelines, while also enhancing patient access through the application of advanced technologies to identify suitable participants and enable remote monitoring.
Who Are The Well-Known Companies In The Clinical Trial Services Market?
Major companies operating in the clinical trial services market are IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON Public Limited Company, WuXi AppTec Co. Ltd., Charles River Laboratories International Inc, Parexel International Corporation, Medpace Holdings Inc, BioClinica Inc, Worldwide Clinical Trials Inc, Medidata Solutions Inc, Precision Medicine Group Inc, Celerion Inc, MakroCare Clinical Research Limited, PCI Pharma Services, Ecron Acunova Limited, PPD LLC., Clinipace Inc., Syntactx LLC., Veristat LLC., AutoCruitment Inc.
Get The Full Clinical Trial Services Market Report:
https://www.thebusinessresearchcompany.com/report/clinical-trial-services-global-market-report
Which Region Represents The Largest Share Of The Clinical Trial Services Market?
North America was the largest region in the clinical trial services market in 2025. North America is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Clinical Trial Services Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/clinical-trial-services-global-market-report
Browse Through More Reports Similar to the Global Clinical Trial Services Market 2026, By The Business Research Company
Clinical Trial Site Management Organizations Market Report 2026
Clinical Trial Management System Market Report 2026
Clinical Trial Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model