Gene Therapy For Inherited Genetic Disorders Market Forecast: How Big Will It Be by 2030?
Uncover key drivers, emerging technologies, and competitive movements shaping the gene therapy for inherited genetic disorders market from 2026–2035 with trusted insights from The Business Research Company
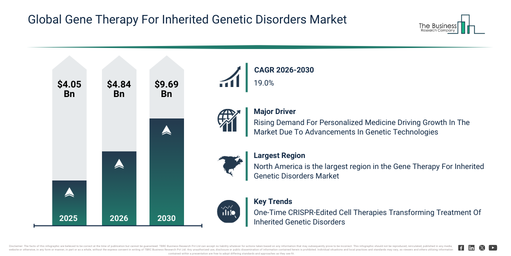
What upper market value could the Gene Therapy For Inherited Genetic Disorders Market reach by 2030 starting from 2026 levels?
The gene therapy for inherited genetic disorders market size has seen rapid expansion in recent years. This market is set to expand from $4.05 billion in 2025 to $4.84 billion in 2026, achieving a compound annual growth rate (CAGR) of 19.3%. Historically, the growth can be linked to aspects such as the limited availability of advanced gene therapy techniques, reliance on conventional treatments for genetic disorders, increasing prevalence of inherited genetic diseases, early clinical research on viral vectors, and rising awareness of rare disease management.
The market for gene therapy targeting inherited genetic disorders is projected to experience swift expansion in the coming years. This market is anticipated to reach $9.69 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 19.0%. This projected growth can be ascribed to several factors, including the advancement of innovative viral vectors and gene editing technologies, the broadening scope of RNA-based treatments, the growing uptake of ex vivo stem cell therapies, increasing financial commitments towards personalized gene therapies, and the enhanced capacity of hospitals and specialty clinics for administering gene therapies. Key trends expected during this period involve the wider application of viral vector-based therapies for uncommon genetic conditions, an increase in ex vivo gene therapy uses, the escalating embrace of RNA-based therapeutics, a heightened emphasis on addressing neurological and ophthalmic ailments, and the incorporation of personalized medicine into gene therapy practices.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27293&type=smp
Which Drivers Are Influencing Long-Term Growth In The Gene Therapy For Inherited Genetic Disorders Market?
The escalating need for personalized medicine is anticipated to fuel the expansion of the gene therapy for inherited genetic disorders market moving forward. Personalized medicine involves customizing medical interventions based on an individual’s genetic profile and personal attributes to achieve optimal patient care. The prevalence of personalized medicine is growing due to breakthroughs in genetic technologies, which facilitate easier and more economical customization of treatments to a person’s distinct genetic composition. This approach enhances gene therapy by tailoring therapies to each patient’s specific genetic makeup, thereby ensuring accurate gene targeting and delivery, while simultaneously improving efficacy and mitigating adverse effects. For illustration, in February 2024, as reported by the Personalized Medicine Coalition, a US-based non-profit organization, the United States Food and Drug Administration (FDA) authorized 16 novel personalized treatments for patients with rare diseases in 2023, an increase from 6 in 2022. These newly approved personalized treatments in 2023 further encompassed seven cancer medications and three for other ailments and conditions. Consequently, the increasing demand for personalized medicine is driving the growth of the gene therapy for inherited genetic disorders market.
Which Segments Are Driving Activity In The Gene Therapy For Inherited Genetic Disorders Market?
The gene therapy for inherited genetic disorders market covered in this report is segmented –
1) By Type: Viral Vector-Based Therapies, Gene Editing Techniques, Ribonucleic Acid-Based Therapies, Ex Vivo Gene Therapy
2) By Application: Inherited Metabolic Disorders, Neurological Disorders, Cardiovascular Disorders, Musculoskeletal Disorders, Ophthalmic Disorders
3) By End-User: Hospitals, Specialty Clinics, Research Institutes, Other End-Users
Subsegments:
1) By Viral Vector-Based Therapies: Adenovirus Vectors, Adeno-Associated Virus (AAV) Vectors, Lentivirus Vectors
2) By Gene Editing Techniques: Zinc Finger Nucleases (ZFNs), Transcription Activator-Like Effector Nucleases (TALENs), Meganucleases
3) By Ribonucleic Acid-Based Therapies: Small Interfering Ribonucleic Acid, Antisense Oligonucleotides (ASOs), Messenger Ribonucleic Acid (mRNA) Therapies
4) By Ex Vivo Gene Therapy: Hematopoietic Stem Cell Gene Therapy, Mesenchymal Stem Cell Gene Therapy, Induced Pluripotent Stem Cell (iPSC) Therapy
How Are New Market Trends Shaping The Landscape Of The Gene Therapy For Inherited Genetic Disorders Market?
Major companies in the gene therapy for inherited genetic disorders market are focused on developing innovative solutions, such as ex-vivo CRISPR/Cas9-based gene-edited cell therapies, to address the increasing demand for one-time, potentially curative treatments. These treatments aim to lessen lifelong dependence on transfusions, repeated hospitalizations, and high chronic-care costs for inherited blood and metabolic disorders. The ex-vivo CRISPR/Cas9 gene-edited cell therapy process involves harvesting a patient’s hematopoietic stem cells, utilizing CRISPR/Cas9 to precisely edit disease-causing genes (or regulatory elements that increase protective fetal hemoglobin), and subsequently reinfusing the modified cells to allow them to engraft and deliver long-term therapeutic benefits. For instance, in December 2023, Vertex Pharmaceuticals, a US-based biopharmaceutical company, received U.S. Food and Drug Administration (FDA) approval and commercial availability for CASGEVY (exagamglogene autotemcel), a CRISPR/Cas9-based, ex-vivo gene-edited cell therapy for sickle cell disease, which is an inherited genetic disorder. CASGEVY is manufactured from a patient’s own hematopoietic stem cells that are genetically altered to increase fetal hemoglobin. The product’s composition centers on autologous stem cells modified with CRISPR/Cas9; its functionality is to raise durable fetal hemoglobin levels that prevent red-blood-cell sickling; unique features include precise genome editing (CRISPR/Cas9) performed ex vivo and a one-time administration with durable effect; its primary application is treating severe sickle cell disease (and related indications under regulatory review), and the main benefits observed in trials are marked reductions in vaso-occlusive crises and transfusion dependence compared with previous standard-of-care.
Which Major Firms Are Strengthening Their Position In The Gene Therapy For Inherited Genetic Disorders Market?
Major companies operating in the gene therapy for inherited genetic disorders market are Sanofi S.A., Novartis AG, Astellas Pharma Inc., Vertex Pharmaceuticals Incorporated, Genentech, Biomarin Pharmaceutical Inc, Sarepta Therapeutics Inc., Krystal Biotech Inc., Rocket Pharmaceuticals Inc., Sangamo Therapeutics Inc., REGENXBIO Inc., Passage Bio Inc., Voyager Therapeutics Inc., Generation Bio Co., Avrobio Inc, Editas Medicine Inc., MeiraGTx Holdings Plc, uniQure N.V., Intellia Therapeutics Inc., Bluebird Bio Inc, 4D Molecular Therapeutics Inc., CRISPR Therapeutics AG
Access The Complete Report For Deeper Market Insights:
Which Region Shows The Strongest Potential For Future Expansion In The Gene Therapy For Inherited Genetic Disorders Market?
North America was the largest region in the gene therapy for inherited genetic disorders market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the gene therapy for inherited genetic disorders market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Gene Therapy For Inherited Genetic Disorders Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27293&type=smp
Browse Through More Reports Similar to the Global Gene Therapy For Inherited Genetic Disorders Market 2026, By The Business Research Company
Asphalt Mixing Plants Market Report 2026
https://www.thebusinessresearchcompany.com/report/asphalt-mixing-plants-global-market-report
Asphalt Pavers Market Report 2026
https://www.thebusinessresearchcompany.com/report/asphalt-pavers-global-market-report
Asphalt Additives Market Report 2026
https://www.thebusinessresearchcompany.com/report/asphalt-additives-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model