Global Gene Therapy For Parkinson’s Disease Industry Insights: Key Trends and Growth Estimates
Uncover key drivers, emerging technologies, and competitive movements shaping the gene therapy for parkinson’s disease market from 2026–2035 with trusted insights from The Business Research Company
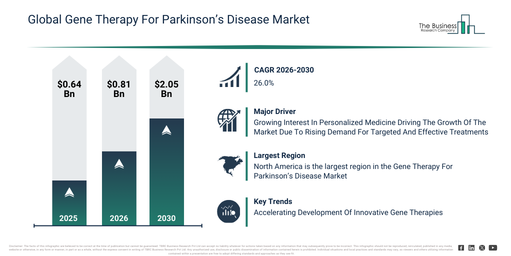
What are the forecasted starting (2026) and ending (2030) market sizes for the Gene Therapy For Parkinson’s Disease Market?
The gene therapy for parkinson’s disease market size has seen significant growth in recent years. This market is anticipated to increase from $0.64 billion in 2025 to $0.81 billion in 2026, achieving a compound annual growth rate (CAGR) of 26.3%. The expansion observed historically stems from progress in viral vector engineering, a greater comprehension of parkinson’s disease pathology, increased funding for neuroscience research, the introduction of enhanced delivery technologies, and successful early clinical trials.
The gene therapy for parkinson’s disease market is projected to undergo significant expansion in the foreseeable future. It is forecasted to achieve a value of $2.05 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 26.0%. This anticipated growth is linked to factors such as increased financial commitments to neurodegenerative disease treatments, the escalating adoption of tailored gene therapy strategies, the advancement of clinical pipelines into late stages, a growing need for treatments that modify the disease’s progression, and enhancements in minimally invasive delivery techniques. Prominent developments expected during this period involve the increasing creation of viral vector-based therapies, a heightened focus on dopamine restoration methods, the expanding application of neurotrophic factor gene delivery, the proliferation of precision intracerebral delivery techniques, and a stronger emphasis on ensuring long-term therapeutic efficacy.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28231&type=smp
What Drivers Are Supporting Technological Adoption In The Gene Therapy For Parkinson’s Disease Market?
The expanding focus on personalized medicine is anticipated to fuel the expansion of the gene therapy for Parkinson’s disease market in the future. Personalized medicine defines a healthcare method designed to customize treatment and prevention plans according to an individual’s genetic profile, way of life, and surroundings. The heightened appeal of personalized medicine stems from an increasing need for specific treatments that boost efficacy and reduce negative side effects. Within personalized medicine, gene therapy for Parkinson’s disease is utilized to provide custom genetic alterations aimed at restoring dopamine function and addressing unique disease pathways in individual patients. As an illustration, in February 2024, the Personalized Medicine Coalition, a US-based non-profit organization, announced that the US Food and Drug Administration (FDA) approved 16 new personalized treatments for rare disease patients in 2023, which represents a significant rise from the 6 approvals in 2022. Consequently, the rising attention to personalized medicine is propelling the growth of the gene therapy for Parkinson’s disease market.
What Segment Types Are Examined In The Gene Therapy For Parkinson’s Disease Market?
The gene therapy for parkinson’s disease market covered in this report is segmented –
1) By Therapy Type: In Vivo Gene Therapy, Ex Vivo Gene Therapy
2) By Vector Type: Viral Vectors, Non-Viral Vectors
3) By Target Gene: Aromatic L-Amino Acid Decarboxylase (AADC), Glial Cell Line-Derived Neurotrophic Factor (GDNF), Neurturin, Other Target Genes
4) By Delivery Method: Intracerebral, Intravenous, Other Delivery Methods
5) By End-User: Hospitals, Specialty Clinics, Research Institutes, Other End-Users
Subsegments:
1) By In Vivo Gene Therapy: Viral Vectors, Non-Viral Vectors
2) By Ex Vivo Gene Therapy: Autologous Cell-Based Gene Therapy, Allogeneic Cell-Based Gene Therapy
What Trends Are Reshaping The Dynamics Of The Gene Therapy For Parkinson’s Disease Market?
Leading companies are active in the gene therapy for Parkinson’s disease market, concentrating on developing more precise treatments, such as regenerative medicine advanced therapy, to enhance neuronal survival, restore dopamine function, and provide long-lasting disease-modifying benefits for patients with Parkinson’s disease. Regenerative medicine advanced therapy (RMAT) refers to a particular United States Food and Drug Administration (FDA) designation granted to regenerative medicine products, including cell therapies, gene therapies, and tissue-engineered products, that are intended to treat, modify, reverse, or cure serious or life-threatening diseases. For instance, in February 2025, Asklepios BioPharmaceutical, Inc. (AskBio), a US-based gene therapy company, introduced AB-1005, an investigational AAV2-GDNF gene therapy that received FDA regenerative medicine advanced therapy (RMAT) designation. This designation, which aims to expedite the development and review of promising therapies, was granted based on data submitted by AskBio, including clinical evidence from its Phase Ib open-label, uncontrolled trial, demonstrating AB-1005’s potential to slow disease progression and improve motor function in Parkinson’s patients.
Who Are The Major Companies Operating In The Gene Therapy For Parkinson’s Disease Market?
Major companies operating in the gene therapy for parkinson’s disease market are Merck & Co. Inc., Bayer AG, Sanofi S.A., Eli Lilly and Company, Biogen Inc., PTC Therapeutics Inc., Zambon S.p.A., Oxford BioMedica plc, Voyager Therapeutics Inc., Insilico Medicine Inc., Spur Therapeutics Inc., Cerevance Inc., MeiraGTx Holdings plc, BlackfinBio Inc., Partner Therapeutics Inc., Kenai Therapeutics Inc., CureSen Therapeutics Inc., BioVie Inc., Clexio Biosciences Ltd., Tetraneuron S.L.
Access The Complete Report For Deeper Market Insights:
Which Region Is Anticipated To See The Fastest Growth In The Gene Therapy For Parkinson’s Disease Market?
North America was the largest region in the gene therapy for parkinson’s disease market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the gene therapy for parkinson’s disease market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Gene Therapy For Parkinson’s Disease Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28231&type=smp
Browse Through More Reports Similar to the Global Gene Therapy For Parkinson’s Disease Market 2026, By The Business Research Company
Genes Therapy Market Report 2026
https://www.thebusinessresearchcompany.com/report/genes-therapy-global-market-report
Anti Parkinson Drugs Market Report 2026
https://www.thebusinessresearchcompany.com/report/anti-parkinson-drugs-global-market-report
Nucleic Acid Based Gene Therapy Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model